Trastuzumab 440 mg Lyophilized Powder for IV Infusion
Trastuzumab 440 mg Lyophilized Powder for IV Infusion Specification
- Indication
- HER2-positive breast cancer, HER2-positive metastatic gastric cancer
- Pacakaging (Quantity Per Box)
- 1 vial per box
- Origin of Medicine
- India
- Salt Composition
- Trastuzumab 440 mg
- Brand Name
- Trastuzumab
- Packaging Type
- Single-dose vial
- Dosage Form
- Injection, lyophilized powder
- Life Span
- 24 months from date of manufacture
- Drug Type
- Biological / Antibody therapy
- Ingredients
- Trastuzumab (recombinant humanized monoclonal antibody) as lyophilized powder
- Physical Form
- Lyophilized powder for IV infusion
- Function
- Antineoplastic (targeted therapy against HER2-overexpressing cancers)
- Recommended For
- Patients with HER2-positive breast cancer and metastatic gastric cancer
- Dosage
- 440 mg per vial; dosing as per physicians protocol, often initial 8 mg/kg IV infusion followed by 6 mg/kg every 3 weeks
- Dosage Guidelines
- Dilute with sterile water before infusion; administered by healthcare professional
- Suitable For
- Adults
- Quantity
- 1 vial
- Storage Instructions
- Store at 2C to 8C (36F to 46F), protect from light, do not freeze
- Contraindications
- Known hypersensitivity to trastuzumab or its excipients
- Cautions
- Monitor for infusion and cardiac-related reactions
- Infusion Time
- Initial dose over 90 minutes; subsequent doses may be given over 30 minutes if tolerated
- Regulatory Status
- Prescription medicine, Schedule H
- Inactive Ingredients
- L-histidine, L-histidine hydrochloride, trehalose, polysorbate 20
- Appearance After Reconstitution
- Clear to opalescent, colorless to pale yellow solution
- Administration Route
- Intravenous infusion (not for IV push or bolus)
- Concentration After Reconstitution
- 21 mg/mL
- Box Contents
- 1 single-use vial + 20 mL bacteriostatic water for injection
- Therapeutic Class
- Monoclonal antibody, Antineoplastic
- Prescription Required
- Yes
- Reconstitution
- Should be reconstituted with 20 mL bacteriostatic water for injection
Trastuzumab 440 mg Lyophilized Powder for IV Infusion Trade Information
- Minimum Order Quantity
- 100 Packs
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 10 Days
- Main Domestic Market
- All India
About Trastuzumab 440 mg Lyophilized Powder for IV Infusion
Trastuzumab 440 mg: Application in Precision Oncology
Trastuzumab 440 mg Lyophilized Powder for IV Infusion is expertly used as a targeted monoclonal antibody therapy in oncology. Its application is paramount for patients diagnosed with HER2-positive breast and metastatic gastric cancers. Administered exclusively in clinical settings under strict medical supervision, it is pivotal in hospital oncology wards, cancer specialty clinics, and advanced treatment centers-delivering specialized, patient-focused care across India and globally.
Reliable Supply and Streamlined Delivery Across India
With outstanding supply ability, Trastuzumab 440 mg is available to meet rising oncology demands with minimal expenditure on logistics. Delivery time is prompt, maintaining the medicine's optimal condition and valuation, adhering to stringent cold chain protocols. The main domestic market spans major metropolitan and tier-1 cities across India, ensuring reliable access to this transformative biological therapy for healthcare facilities and cancer patients nationwide.
FAQ's of Trastuzumab 440 mg Lyophilized Powder for IV Infusion:
Q: How should Trastuzumab 440 mg Lyophilized Powder be prepared for infusion?
A: Trastuzumab 440 mg should be reconstituted with 20 mL of bacteriostatic water for injection. After gentle mixing, the lyophilized powder forms a clear to opalescent, colorless to pale yellow solution with a concentration of 21 mg/mL, suitable for intravenous infusion.Q: What is the recommended administration route and infusion time for this medication?
A: Trastuzumab is administered only via intravenous infusion-never as an IV push or bolus. The initial dose should be infused over 90 minutes; if tolerated, subsequent doses can be administered over 30 minutes.Q: When is Trastuzumab 440 mg indicated for use?
A: This medicine is indicated for adults with HER2-positive breast cancer and HER2-positive metastatic gastric cancer, providing targeted therapy where overexpression of the HER2 protein is confirmed.Q: Where should Trastuzumab 440 mg be stored before use?
A: The vials must be refrigerated at 2C to 8C (36F to 46F), protected from light. They must not be frozen to preserve efficacy.Q: What precautions should be taken during Trastuzumab infusion?
A: Patients must be monitored closely for infusion-related and cardiac events. Administration should be performed by healthcare professionals in suitable medical facilities to manage any adverse reactions promptly.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharma Injection Category
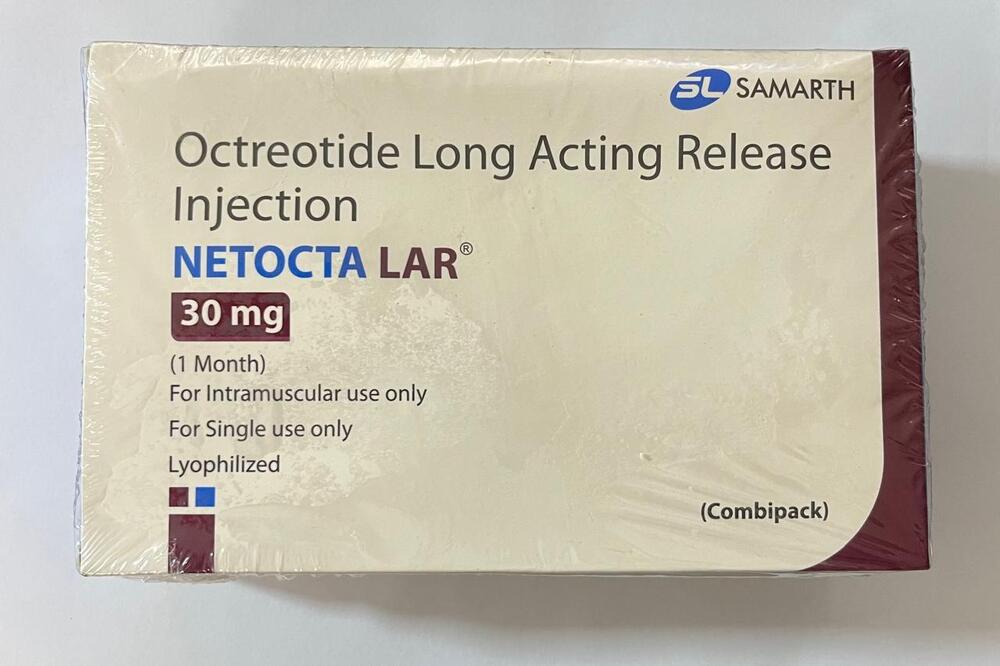
Octreotide Long Acting Release Injection
Minimum Order Quantity : 100 Packs
Dosage Form : Long Acting Release (LAR) Injectable Suspension
Quantity : 1 Vial
Drug Type : Other, Synthetic somatostatin analogue
Life Span : 24 months from manufacture date
Brand Name : Octreotide LAR
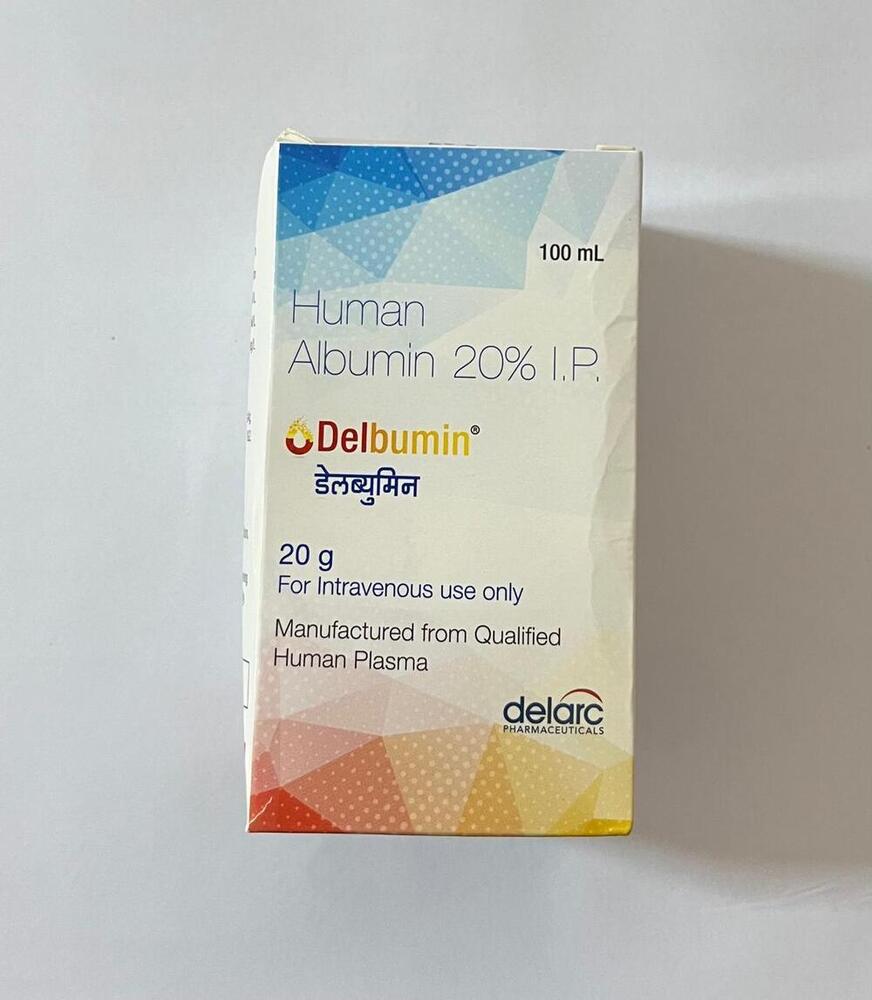
Human Albumin 20g I.P
Minimum Order Quantity : 100 Packs
Dosage Form : Injection
Quantity : 20g
Drug Type : Other, Biological Product
Life Span : 24 months
Brand Name : Human Albumin
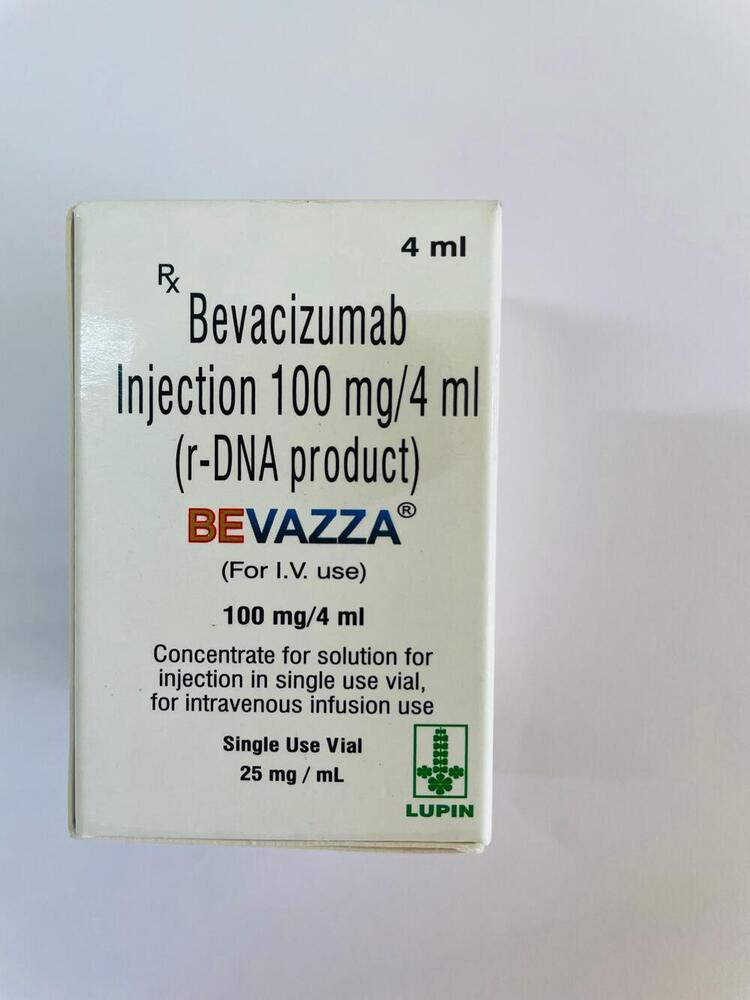
100mg Bevacizumab Injection
Minimum Order Quantity : 100 Packs
Dosage Form : Injection
Quantity : 4 ml Vial
Drug Type : Other, Prescription Drug
Life Span : 24 Months from manufacturing date
Brand Name : Bevacizumab Injection
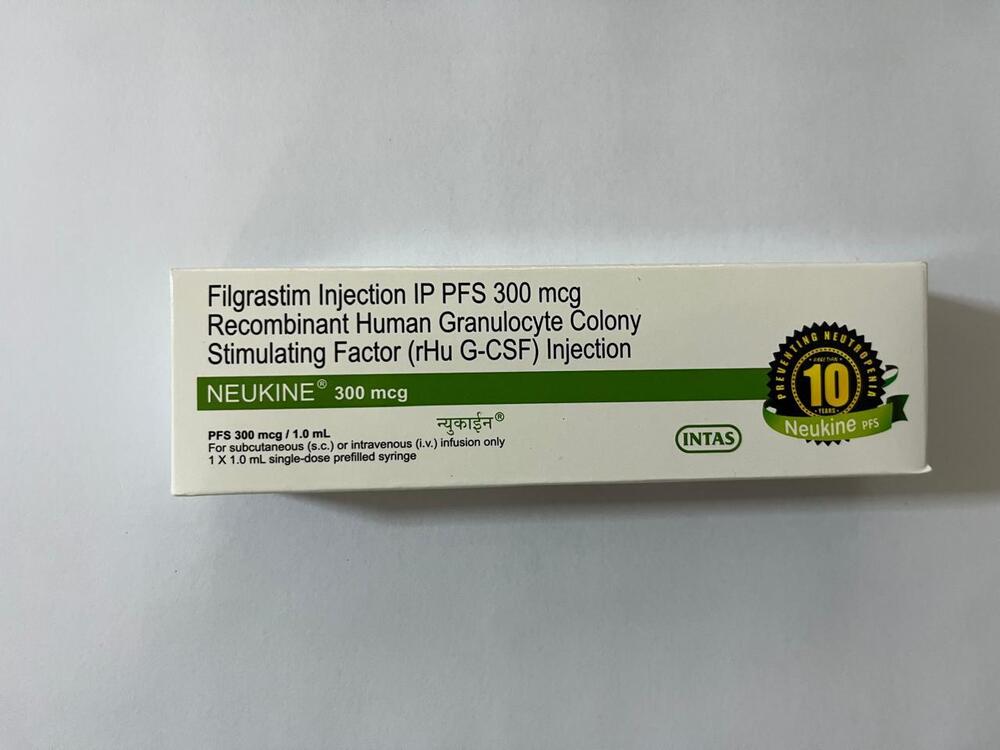
Filgrastim Injection IP PFS 300 Mcg Recombinant Human Granulocyte Colony Stimulation Factor Injection
Minimum Order Quantity : 100 Packs
Dosage Form : Parenteral
Quantity : 1 prefilled syringe
Drug Type : Other, Biological
Life Span : 24 months from manufacturing date
Brand Name : Filgrastim Injection IP
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese