100mg Bevacizumab Injection
100mg Bevacizumab Injection Specification
- Salt Composition
- Bevacizumab
- Dosage Form
- Injection
- Indication
- Metastatic cancer treatment, Macular degeneration, Recurrent Glioblastoma
- Origin of Medicine
- Allopathic
- Packaging Type
- Glass Vial
- Brand Name
- Bevacizumab Injection
- Life Span
- 24 Months from manufacturing date
- Pacakaging (Quantity Per Box)
- 1 Vial per box
- Drug Type
- Prescription Drug
- Ingredients
- Bevacizumab 100mg
- Physical Form
- Liquid Injection
- Function
- Antineoplastic (anticancer) agent, Inhibits angiogenesis
- Recommended For
- Cancer Patients (Colorectal, Lung, Renal Cell Carcinoma, Glioblastoma, Cervical and Ovarian Cancer)
- Dosage
- As directed by physician, typically 5-15 mg/kg every 2-3 weeks
- Dosage Guidelines
- Administered by IV infusion under supervision
- Suitable For
- Adults
- Quantity
- 4 ml Vial
- Storage Instructions
- Store between 2C to 8C, Do not freeze
- Shelf Life after Opening
- Use immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user.
- Contraindications
- Known hypersensitivity to Bevacizumab or any excipients
- Side Effects
- Proteinuria, hypertension, bleeding, GI perforations (see physician for full profile)
- Warning
- Use only under medical supervision, not for use in pregnancy or breastfeeding unless advised
- Preservative
- Preservative free
- Market Authorization Holder
- As per regulatory approval
- Therapeutic Class
- Monoclonal Antibody (Anti-VEGF)
- Route of Administration
- Parenteral
- Concentration
- 25 mg/ml
- ATC Code
- L01XC07
- Method of Administration
- Intravenous use only (IV infusion)
- Appearance
- Clear to slightly opalescent, colorless to pale brownish-yellow solution
100mg Bevacizumab Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 10 Days
- Main Domestic Market
- All India
About 100mg Bevacizumab Injection
Method of Administration and Expanded Applications
100mg Bevacizumab Injection is administered via intravenous infusion, ensuring direct and effective delivery to the bloodstream. While primarily indicated for metastatic cancers such as colorectal, lung, and ovarian, its application has been extended to conditions like macular degeneration and recurrent glioblastoma. Distinct features include a preservative-free, colorless to pale brownish-yellow, clear solution, housed in a robust 4 ml glass vial. This thoughtful design makes it ideal for precise dosing and ensures product integrity throughout its lifespan.
Sample Availability, Port, and Payment Solutions
Samples of the 100mg Bevacizumab Injection are available for evaluation, with supplies handed over efficiently through reliable distribution from our designated FOB port in India. Flexible payment terms can be discussed to suit your proposal amount, ensuring a seamless transaction experience. Our commitment is reflected in streamlined processes, from sample requests to finalized orders, providing robust support and prompt supply to meet medical requirements. Request your sample and secure your needed stock through straightforward commercial arrangements.
FAQ's of 100mg Bevacizumab Injection:
Q: How is 100mg Bevacizumab Injection administered?
A: The injection is administered intravenously (IV infusion) by a trained healthcare professional, ensuring the medication is delivered directly into the bloodstream for optimal therapeutic effects.Q: What conditions can Bevacizumab Injection be used to treat?
A: Bevacizumab Injection is indicated for the treatment of metastatic cancers such as colorectal, lung, renal cell carcinoma, glioblastoma, cervical, and ovarian cancer. It is also used for macular degeneration and recurrent glioblastoma.Q: When should Bevacizumab Injection not be used?
A: It should not be used in patients with known hypersensitivity to Bevacizumab or any of its excipients. It is also not recommended during pregnancy or breastfeeding, unless specifically advised by a physician.Q: Where should Bevacizumab Injection be stored?
A: The injection must be stored between 2C to 8C and should never be frozen. Proper refrigeration preserves product efficacy and safety until administration.Q: What is the process for obtaining a sample or placing an order?
A: Contact us with your requirements. After discussing the proposal amount and supply needs, samples can be handed over at the designated FOB port, with flexible payment terms arranged to facilitate your acquisition.Q: How does Bevacizumab Injection benefit cancer patients?
A: Bevacizumab works as an antineoplastic agent inhibiting angiogenesis, blocking the formation of new blood vessels that tumors need for growth, thereby restricting cancer progression under expert medical supervision.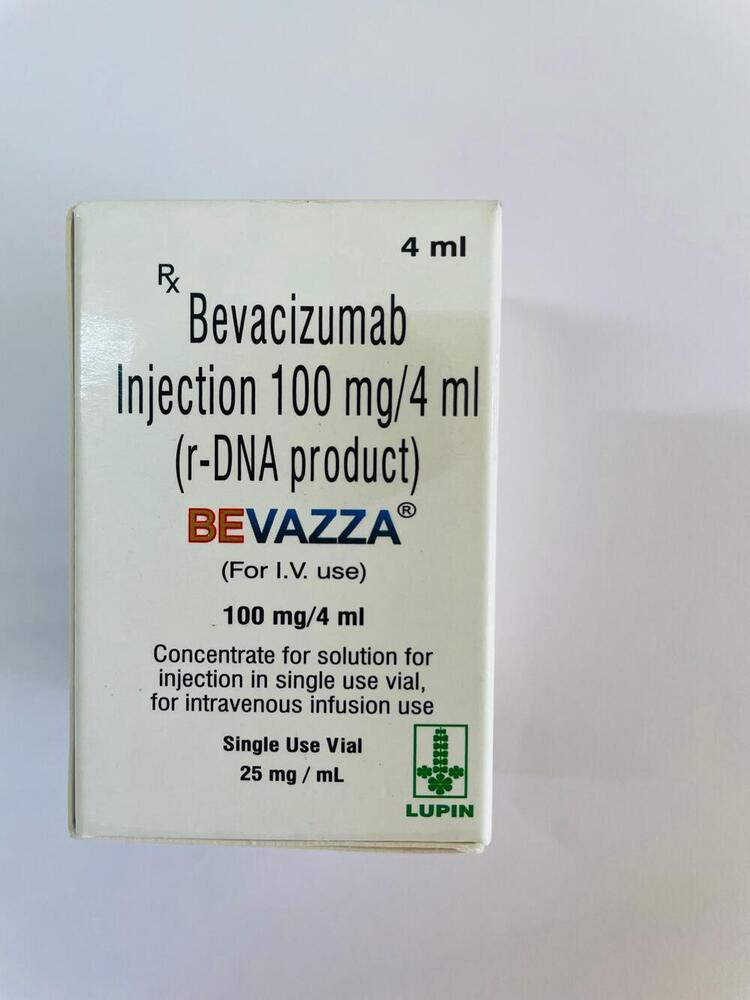

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharma Injection Category
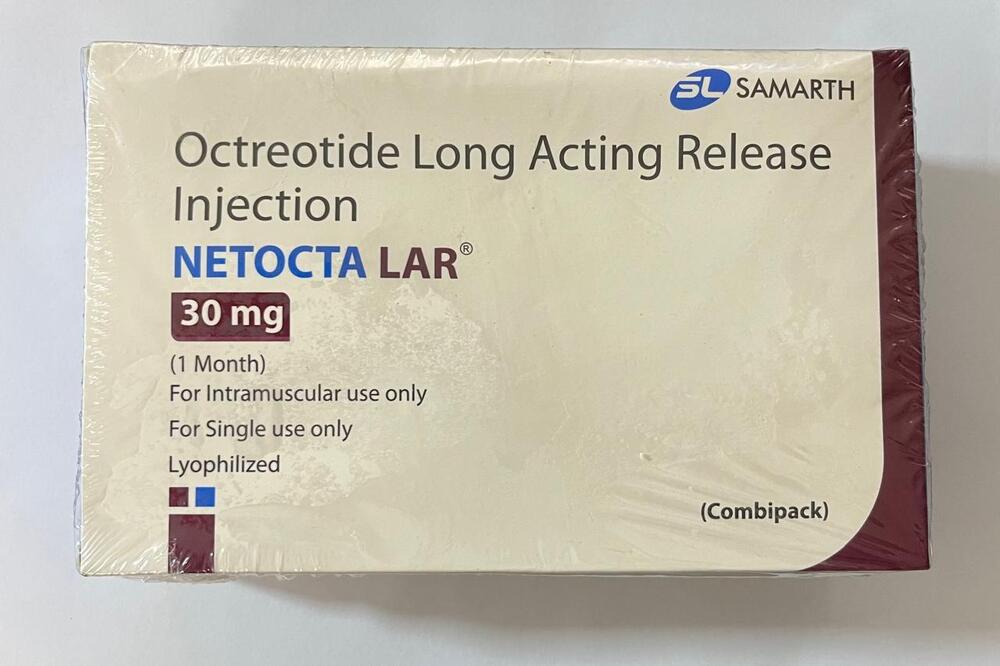
Octreotide Long Acting Release Injection
Minimum Order Quantity : 100 Packs
Dosage Form : Long Acting Release (LAR) Injectable Suspension
Salt Composition : Octreotide Acetate
Indication : Acromegaly, neuroendocrine tumors, carcinoid syndrome
Physical Form : Other, Injection
Origin of Medicine : India
Trastuzumab 440 mg Lyophilized Powder for IV Infusion
Minimum Order Quantity : 100 Packs
Dosage Form : Injection, lyophilized powder
Salt Composition : Trastuzumab 440 mg
Indication : HER2positive breast cancer, HER2positive metastatic gastric cancer
Physical Form : Other, Lyophilized powder for IV infusion
Origin of Medicine : India
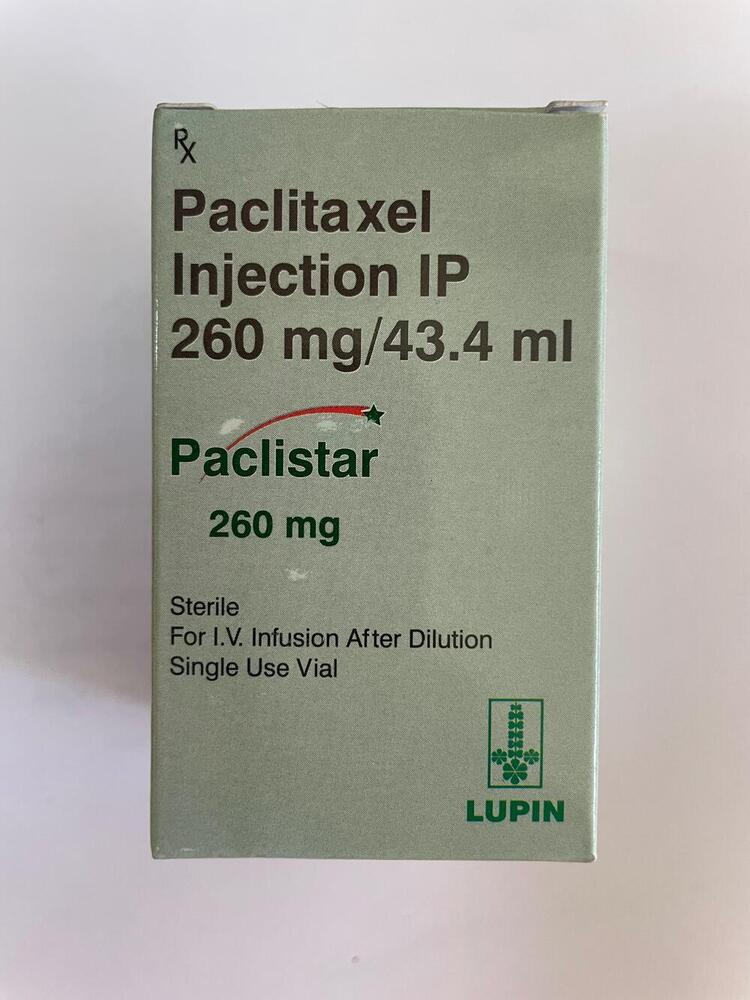
260MG Paclitaxel Injection IP
Minimum Order Quantity : 100 Packs
Dosage Form : Injection
Salt Composition : Paclitaxel
Indication : Ovarian cancer, Breast cancer, Nonsmall cell lung cancer and AIDSrelated Kaposis sarcoma
Physical Form : Liquid
Origin of Medicine : India
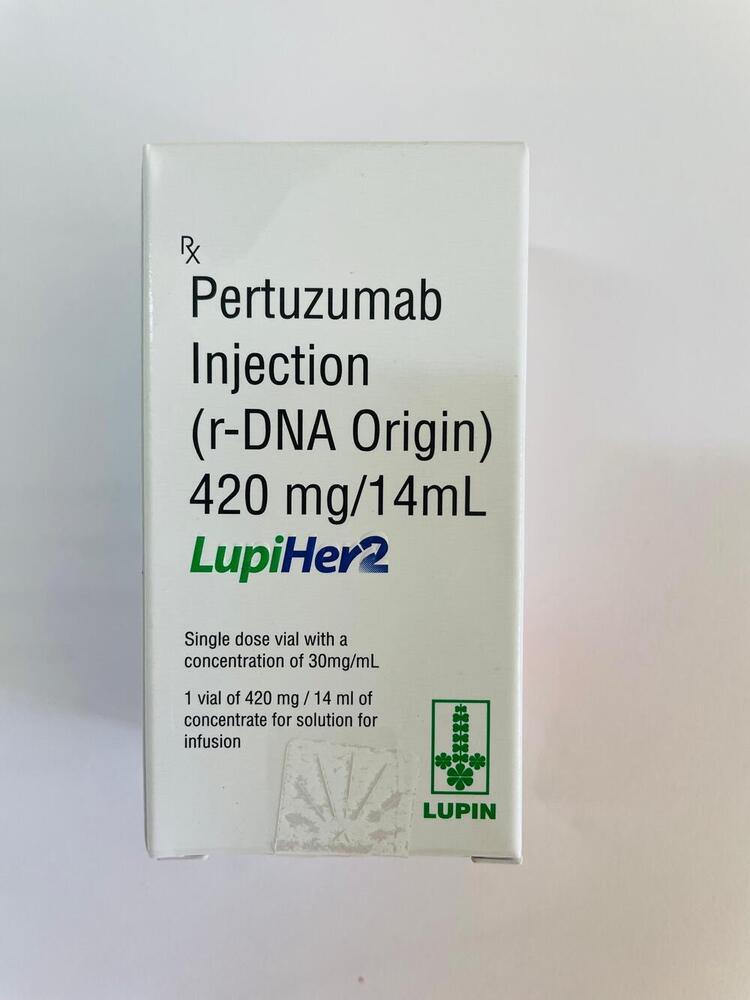
Pertuzumab Injection
Minimum Order Quantity : 100 Packs
Dosage Form : Injection
Salt Composition : Pertuzumab (420 mg)
Indication : Treatment of HER2positive metastatic breast cancer
Physical Form : Liquid
Origin of Medicine : Imported
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese