Filgrastim Injection IP PFS 300 Mcg Recombinant Human Granulocyte Colony Stimulation Factor Injection
Filgrastim Injection IP PFS 300 Mcg Recombinant Human Granulocyte Colony Stimulation Factor Injection Specification
- Salt Composition
- Filgrastim 300 mcg
- Packaging Type
- Pre-Filled Syringe (PFS)
- Indication
- Neutropenia, bone marrow transplantation, and chemotherapy-induced neutropenia
- Dosage Form
- Parenteral
- Brand Name
- Filgrastim Injection IP
- Pacakaging (Quantity Per Box)
- 1 syringe per box
- Origin of Medicine
- India
- Life Span
- 24 months from manufacturing date
- Drug Type
- Biological
- Ingredients
- Recombinant Human Granulocyte Colony Stimulating Factor (Filgrastim) 300 mcg
- Physical Form
- Injection
- Function
- Boost white blood cell count
- Recommended For
- Prevention of neutropenia in patients undergoing chemotherapy
- Dosage
- As directed by the physician
- Dosage Guidelines
- Administer subcutaneously or intravenously as per medical supervision
- Suitable For
- Adults and pediatric patients as prescribed
- Quantity
- 1 pre-filled syringe
- Storage Instructions
- Store between 2C to 8C. Do not freeze.
- Prescription Required
- Yes
- Common Side Effects
- Bone pain, fever, injection site reactions
- Marketed By
- As mentioned on packaging
- Manufacturing License No.
- As mentioned on packaging
- Route of Administration
- Subcutaneous or intravenous use
- Appearance
- Clear, colorless solution
- Precautions
- Not to be used in patients with hypersensitivity to filgrastim or E. coli derived proteins
- Use in Pregnancy and Lactation
- Use only if prescribed by your doctor
Filgrastim Injection IP PFS 300 Mcg Recombinant Human Granulocyte Colony Stimulation Factor Injection Trade Information
- Minimum Order Quantity
- 100 Packs
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 5000 Packs Per Month
- Delivery Time
- 10 Days
- Main Domestic Market
- All India
About Filgrastim Injection IP PFS 300 Mcg Recombinant Human Granulocyte Colony Stimulation Factor Injection
Sterling Features and Applications
Filgrastim Injection IP PFS 300 Mcg boasts a clear, colorless solution with champion performance in neutropenia management. Its unparalleled formulation, featuring Recombinant Human Granulocyte Colony Stimulating Factor, is indicated for use in patients undergoing chemotherapy, bone marrow transplantation, or those at risk for chemotherapy-induced neutropenia. With a pre-filled syringe ensuring ease of use and precise administration, this parenteral injection is ideal for both adults and pediatric patients as directed by a physician.
Export Markets, Supply, and Ordering Process
Recognized in major export markets, Filgrastim Injection IP PFS 300 Mcg has robust supply ability and flexible sample policy for distributors and healthcare providers. Order processing is seamless and tailored to meet bulk requirements, with attractive proposal amounts linked to list prices and quantity per box. Reliable delivery ensures medical facilities receive products promptly, supporting uninterrupted patient care.
FAQ's of Filgrastim Injection IP PFS 300 Mcg Recombinant Human Granulocyte Colony Stimulation Factor Injection:
Q: How should Filgrastim Injection IP PFS 300 Mcg be administered?
A: Filgrastim Injection IP PFS 300 Mcg should be administered either subcutaneously or intravenously, strictly as per the dosage and guidelines provided by your physician.Q: What is the primary benefit of using Filgrastim Injection IP PFS 300 Mcg?
A: The main benefit is the prevention and management of neutropenia, particularly in patients undergoing chemotherapy or bone marrow transplantation, by boosting white blood cell count.Q: When should Filgrastim Injection IP PFS 300 Mcg be used during pregnancy or lactation?
A: This injection should only be used during pregnancy or lactation if prescribed by your doctor, after careful consideration of the potential benefits and risks.Q: Where should Filgrastim Injection IP PFS 300 Mcg be stored?
A: Store the pre-filled syringe in a refrigerator between 2C and 8C. It should not be frozen and must be kept away from light until use.Q: What precautions should be taken before administering this injection?
A: Filgrastim Injection IP PFS 300 Mcg must not be used in patients with known hypersensitivity to filgrastim or E. coli-derived proteins. Consult your healthcare provider for a full list of precautions.Q: How many injections are included per box of Filgrastim Injection IP PFS 300 Mcg?
A: Each package contains one pre-filled syringe, making dosing straightforward and accurate.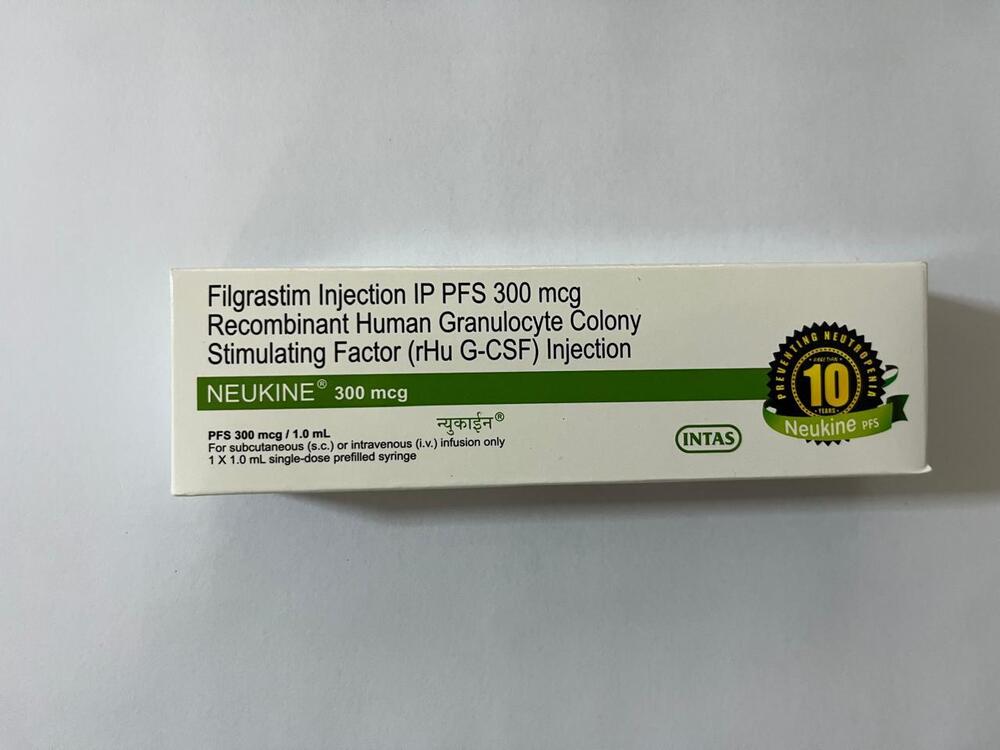

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Pharma Injection Category
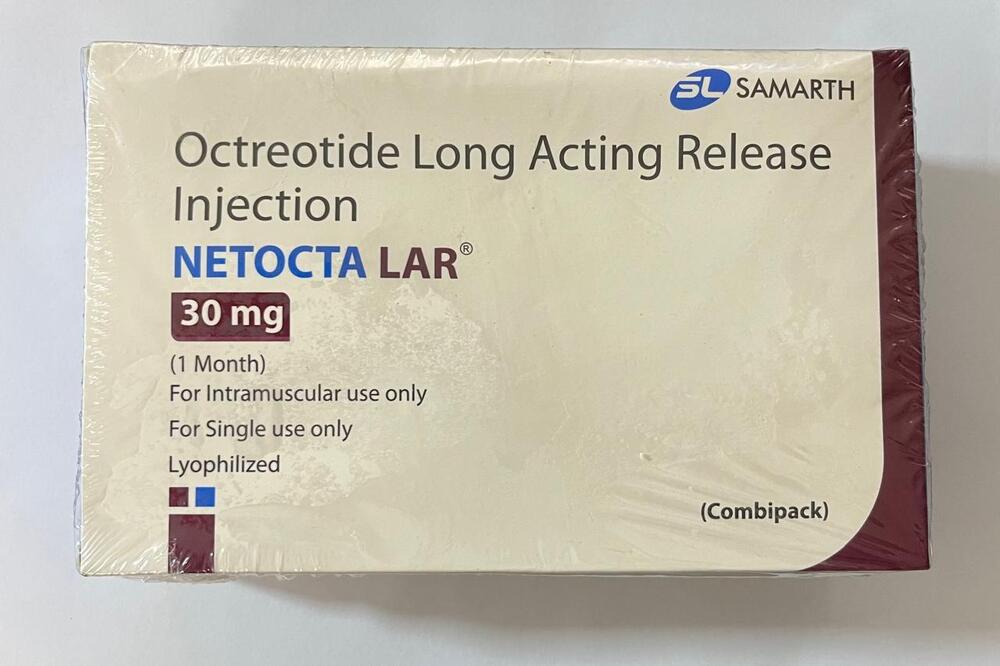
Octreotide Long Acting Release Injection
Minimum Order Quantity : 100 Packs
Pacakaging (Quantity Per Box) : 1 vial with diluent and injection kit
Storage Instructions : Store at 2C to 8C (Refrigerate, do not freeze). Protect from light.
Origin of Medicine : India
Life Span : 24 months from manufacture date
Indication : Acromegaly, neuroendocrine tumors, carcinoid syndrome
500MG Rituximab Injection IP
Minimum Order Quantity : 100 Packs
Pacakaging (Quantity Per Box) : 1 vial per box
Storage Instructions : Store at 2C 8C. Do not freeze. Protect from light.
Origin of Medicine : India
Life Span : 24 months from manufacturing date
Indication : For the treatment of certain types of cancer and autoimmune diseases
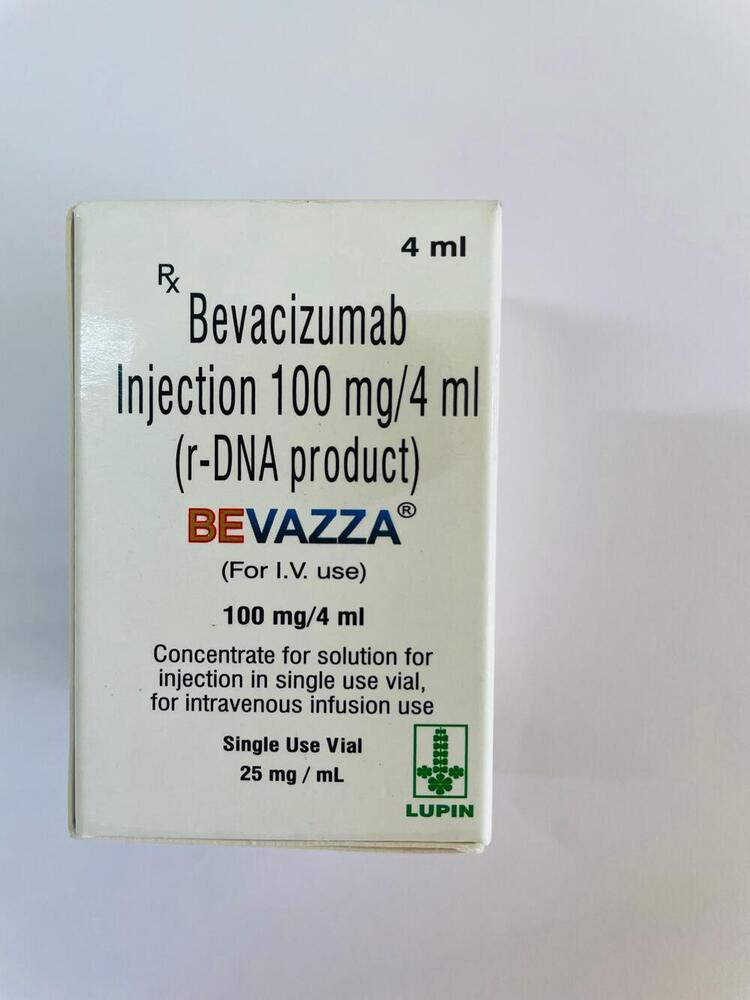
100mg Bevacizumab Injection
Minimum Order Quantity : 100 Packs
Pacakaging (Quantity Per Box) : 1 Vial per box
Storage Instructions : Store between 2C to 8C, Do not freeze
Origin of Medicine : Allopathic
Life Span : 24 Months from manufacturing date
Indication : Metastatic cancer treatment, Macular degeneration, Recurrent Glioblastoma
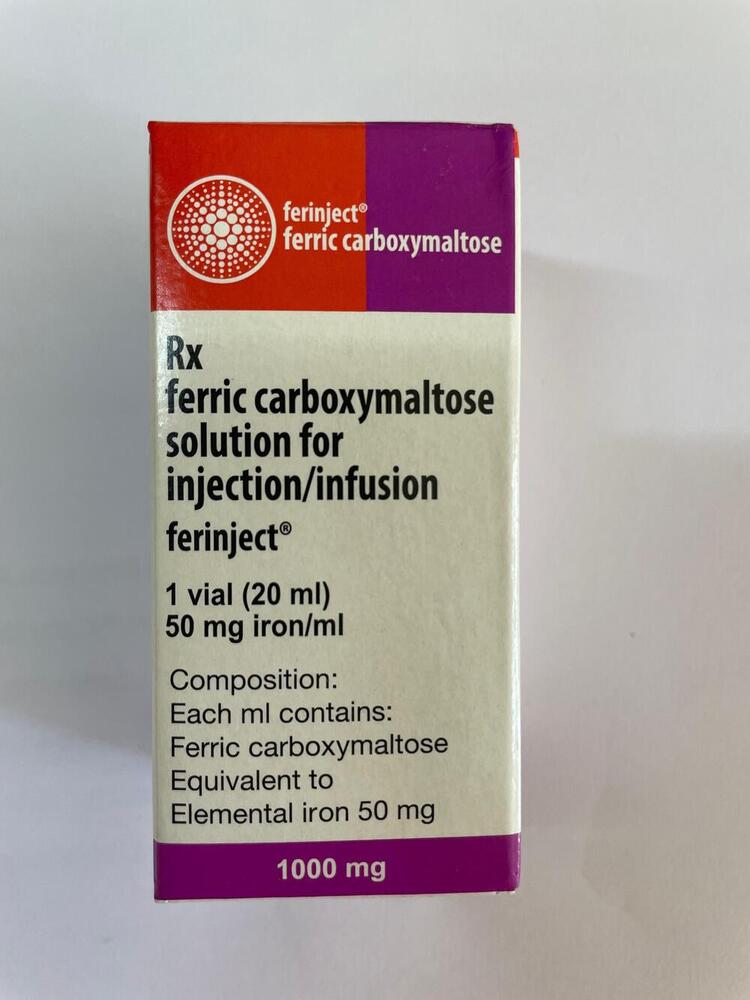
RX Ferric Carboxymaltose Solution For Injection
Minimum Order Quantity : 100 Packs
Pacakaging (Quantity Per Box) : 1 vial per box
Storage Instructions : Store below 25C, protect from light
Origin of Medicine : India
Life Span : 24 months from manufacturing
Indication : Treatment of iron deficiency where oral iron therapy is ineffective or cannot be used
 Send Inquiry
Send Inquiry Send Inquiry
Send Inquiry

 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese